Samsung Biologics at AACR 2026: Accelerating cancer drug development with organoid and bispecific capabilities

The American Association for Cancer Research (AACR) Annual Meeting is one of the top three global oncology conferences, bringing together key stakeholders to share the latest scientific and clinical advancements. At this year’s meeting, held in San Diego from April 17 to 22, Samsung Biologics showcased its organoid services and bispecific antibody technologies while strengthening engagement with potential clients.

Alexis Santana, Director of Organoid Sales at Samsung Biologics, hosted a speaker session to introduce Samsung Organoids, a human-relevant preclinical platform for early compound screening. With high similarity to patient tumor tissue, expandable immune cells, and rapid screening timelines, the platform helps reduce drug candidate attrition prior to clinical trials. By incorporating organoid-based screening early in the drug development process, Samsung Biologics helps clients accelerate development and mitigate risks.
Regulatory agencies are encouraging the use of New Approach Methodologies (NAMs), including organoid-based data, while the U.S. Food and Drug Administration (FDA) Modernization Act 2.0 has eliminated the federal mandate for animal testing in drug development, opening the door to non-animal alternatives. Recently, the FDA issued draft guidance that provides a validation framework for NAMs to improve predictive toxicology in humans and further reduce reliance on animal testing.

Overcoming the limitations of traditional 2D cell lines, Samsung Organoids enables clinically relevant drug screening, by leveraging patient treatment information, multimodal datasets, and next-generation data derived from sequencing. The platform is designed to help identify lead candidates and accelerate preclinical development. By combining screening results with genomic and clinical data, Samsung Biologics supports preclinical development, identifies biomarkers of response or resistance, and generates insights to inform patient selection and treatment strategies.

Seahee Kim, Director of Organoids Technology, presented a poster highlighting Samsung Organoids, demonstrating that its patient-derived organoids (PDOs) platform recapitulates tumor mutations and drug responses, achieving 85% concordance with treatment outcomes and delivering multi-omics insights into sensitivity and resistance. Seahee Kim showcased an analysis of 24 PDOs generated from fresh tumor tissue linked to the anonymized clinical records of patients with colorectal cancer at Samsung Medical Center. The results showed that Samsung Organoids improves clinical predictability by precisely assessing drug responses and supports biomarker discovery through the integration of clinical and multi-omics data.
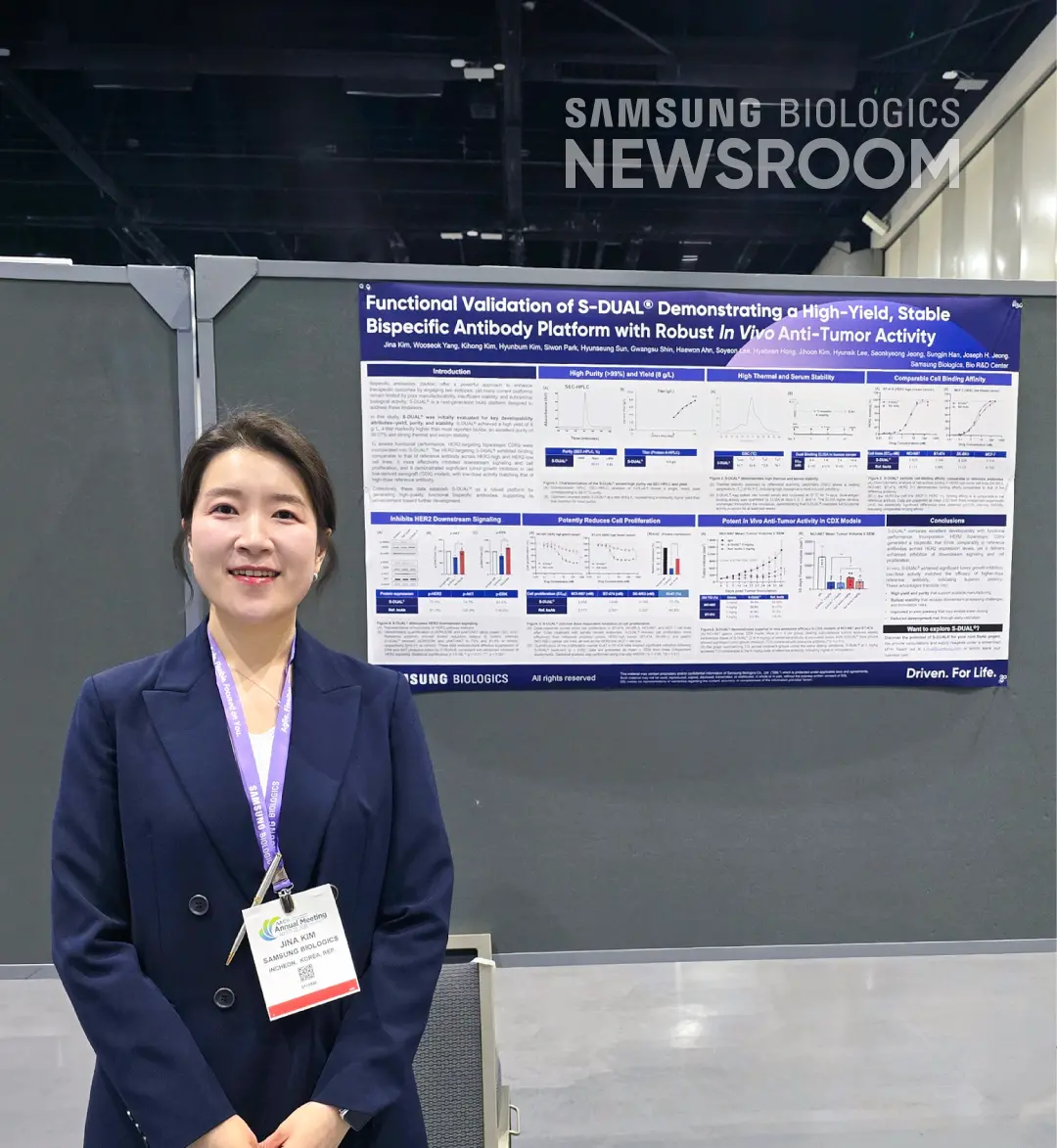
Jina Kim, Director of Antibody Technology Discovery, presented a poster highlighting Samsung Biologics’ bispecific antibody platform, S-DUAL®, which combines developability attributes with functional performance assessments. Jina Kim presented a study demonstrating that S-DUAL® achieved high yield and purity, with titers markedly exceeding those of most reported bispecific antibodies. The researchers assessed functional performance using HER2-targeting biparatopic complementarity-determining regions (CDRs) incorporated into S-DUAL® as a case study to demonstrate superior performance of the platform. The HER2-targeting S-DUAL® demonstrated binding abilities comparable to those of reference antibodies across HER2 expression levels while enhancing the inhibition of downstream signaling and cell proliferation.
At the AACR Annual Meeting 2026, Samsung Biologics underscored its dedication to transforming oncology drug development with human-relevant preclinical models. By showcasing its Samsung Organoids portfolio and the high-yield, high-purity S-DUAL® bispecific antibody platform, the company demonstrated how its integrated services can compress discovery timelines, boost predictive accuracy, and lower attrition — bringing safer, lifesaving solutions to patients more efficiently.

The American Association for Cancer Research (AACR) Annual Meeting is one of the top three global oncology conferences, bringing together key stakeholders to share the latest scientific and clinical advancements. At this year’s meeting, held in San Diego from April 17 to 22, Samsung Biologics showcased its organoid services and bispecific antibody technologies while strengthening engagement with potential clients.

Alexis Santana, Director of Organoid Sales at Samsung Biologics, hosted a speaker session to introduce Samsung Organoids, a human-relevant preclinical platform for early compound screening. With high similarity to patient tumor tissue, expandable immune cells, and rapid screening timelines, the platform helps reduce drug candidate attrition prior to clinical trials. By incorporating organoid-based screening early in the drug development process, Samsung Biologics helps clients accelerate development and mitigate risks.
Regulatory agencies are encouraging the use of New Approach Methodologies (NAMs), including organoid-based data, while the U.S. Food and Drug Administration (FDA) Modernization Act 2.0 has eliminated the federal mandate for animal testing in drug development, opening the door to non-animal alternatives. Recently, the FDA issued draft guidance that provides a validation framework for NAMs to improve predictive toxicology in humans and further reduce reliance on animal testing.

Overcoming the limitations of traditional 2D cell lines, Samsung Organoids enables clinically relevant drug screening, by leveraging patient treatment information, multimodal datasets, and next-generation data derived from sequencing. The platform is designed to help identify lead candidates and accelerate preclinical development. By combining screening results with genomic and clinical data, Samsung Biologics supports preclinical development, identifies biomarkers of response or resistance, and generates insights to inform patient selection and treatment strategies.
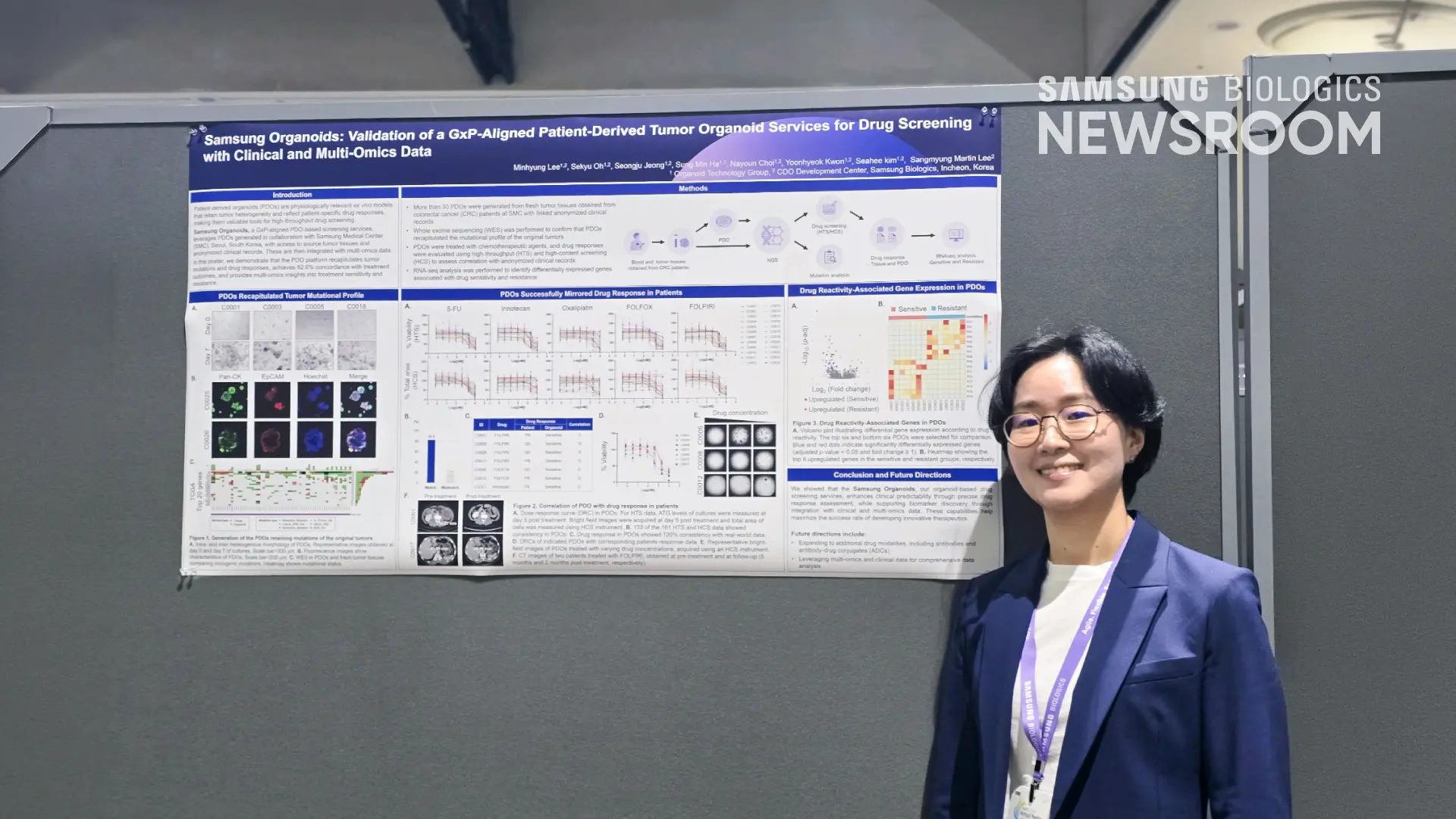
Seahee Kim, Director of Organoids Technology, presented a poster highlighting Samsung Organoids, demonstrating that its patient-derived organoids (PDOs) platform recapitulates tumor mutations and drug responses, achieving 85% concordance with treatment outcomes and delivering multi-omics insights into sensitivity and resistance. Seahee Kim showcased an analysis of 24 PDOs generated from fresh tumor tissue linked to the anonymized clinical records of patients with colorectal cancer at Samsung Medical Center. The results showed that Samsung Organoids improves clinical predictability by precisely assessing drug responses and supports biomarker discovery through the integration of clinical and multi-omics data.

Jina Kim, Director of Antibody Technology Discovery, presented a poster highlighting Samsung Biologics’ bispecific antibody platform, S-DUAL®, which combines developability attributes with functional performance assessments. Jina Kim presented a study demonstrating that S-DUAL® achieved high yield and purity, with titers markedly exceeding those of most reported bispecific antibodies. The researchers assessed functional performance using HER2-targeting biparatopic complementarity-determining regions (CDRs) incorporated into S-DUAL® as a case study to demonstrate superior performance of the platform. The HER2-targeting S-DUAL® demonstrated binding abilities comparable to those of reference antibodies across HER2 expression levels while enhancing the inhibition of downstream signaling and cell proliferation.
At the AACR Annual Meeting 2026, Samsung Biologics underscored its dedication to transforming oncology drug development with human-relevant preclinical models. By showcasing its Samsung Organoids portfolio and the high-yield, high-purity S-DUAL® bispecific antibody platform, the company demonstrated how its integrated services can compress discovery timelines, boost predictive accuracy, and lower attrition — bringing safer, lifesaving solutions to patients more efficiently.
Share article
Related Content