Timeline
From CLD to IND Filing in 9 Months
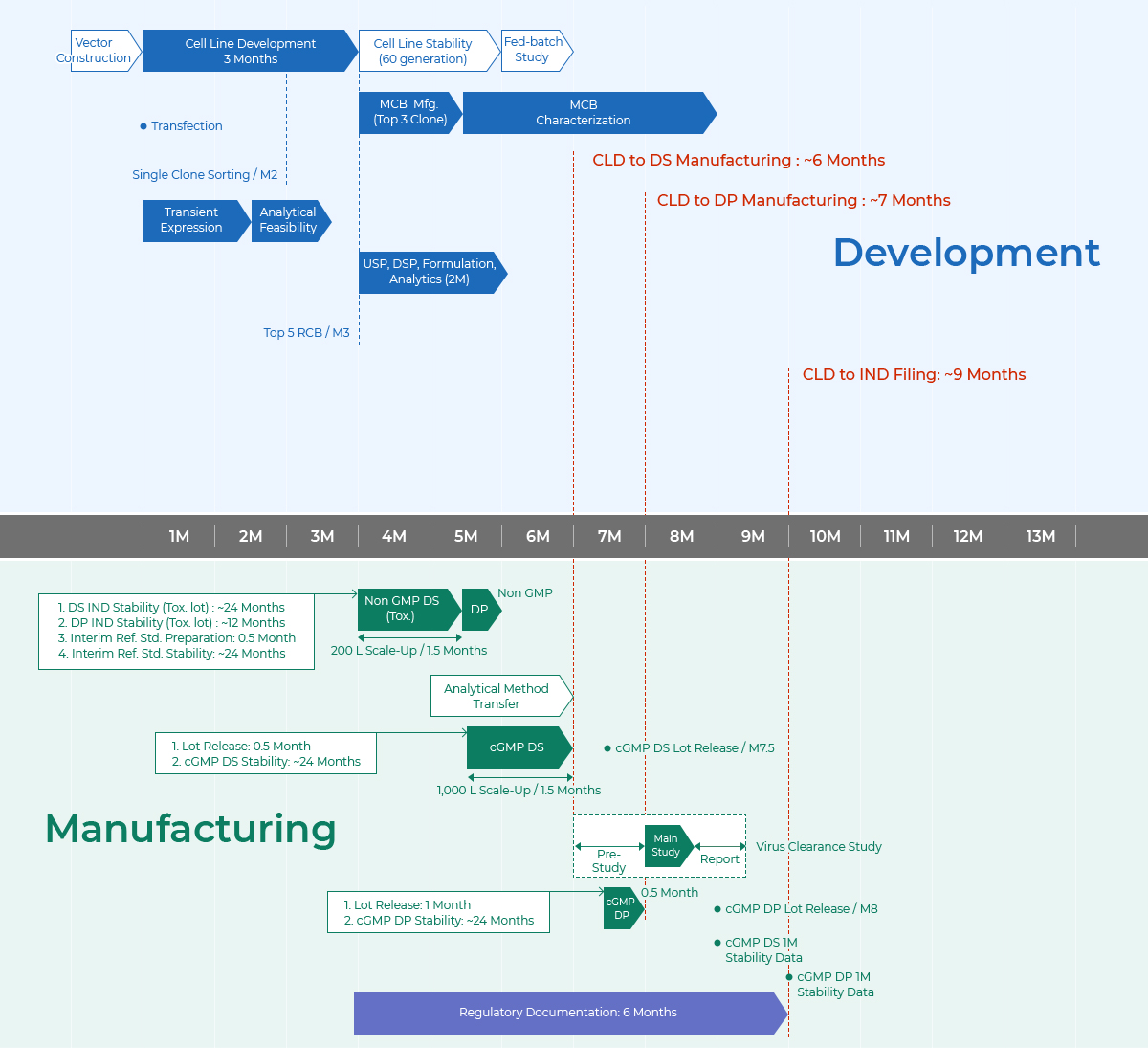
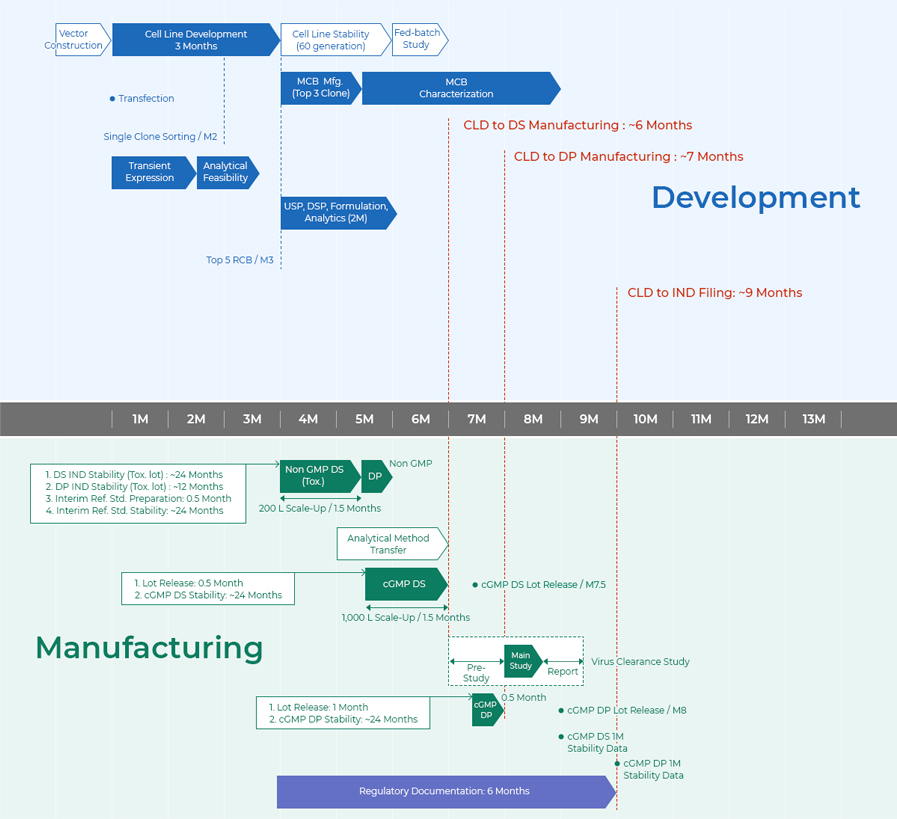
- Development
-
- 0M
- Vector Construction
- 1M ~ 3M
- Cell Line Development 3 months Transfection Single Clone Sorting 2months
- 4M ~ 7M
- Cell Line Stability 2.5 months Pre-Bank Testing(MCB Prep. MCB Characterization) MCB Manufacturing / 4months SBL Process Platform USP[Clone Feasibility(2L Run), Tox(200L) ] DSP Formulation Analytical Performance Test Tox DP
- Manufacturing
-
- 6M ~ 9M
- IRS Qualification : 1.5 months IRS Characterization : 3 months Analytical Method Transfers 3 months CGMP DS(1,000L Scale-Up / 1.5 months) 1. Lot Releases : 1 month 2. CGMP DS Stability : ~ 24 months CLD to DS Manufacturing: ~ 9 months
- 10M ~ 12M
- CGMP DS Lot Release 10 months CGMP DP 0.6 month 1. Lot Releases : 1 month 2. CGMP DP Stability : ~ 24 months CLD to DP Manufacturing: ~ 10.5 months CGMP DP Lot RElease 11.5 months pre-Study Main Study Report Virus Clearance Study 2.5 months
- 13M
- CLD to IND Filing: ~ 12.5 months Regulatory Documentation : 6 months CGMP DS 1M Stability Data CGMP DP 1M Stability Data
MCB : Master Cell Bank
zoom in